Breaking Azeotropes with Confidence Using PBI Membranes
Traditional methods of separating water from industrial solvents are energy-intensive and limited by physical constraints on purity.
Distillation, the most common method, is often restricted by azeotropes, high boiling points, and solvent instability. These challenges frequently require costly workarounds such as ternary distillation.
Pervaporation offers a more efficient alternative. Instead of boiling and condensing, it draws water through a selective membrane, evaporates it under vacuum, and collects it as vapor.
The result: lower energy consumption, higher purity output, and a reliable path around azeotropes.
Why Pervaporation Outperforms Distillation
Pervaporation eliminates the need for high heat input and complex distillation setups.
- Bypasses azeotropic limitations
- Reduces energy consumption
- Improves final product purity
- Enables stable separation at lower temperatures
This makes it especially valuable in solvent dehydration processes where efficiency and purity are critical.
The PBI Advantage in Harsh Solvent Systems
Membrane durability is critical in pervaporation systems. Many polymers degrade under solvent exposure, heat, or aggressive chemistry.
Celazole® PBI solves this through exceptional thermal and chemical stability.
- Thermal strength: Maintains stability with a 427°C glass transition temperature
- Solvent resistance: Withstands ethanol, acetone, hydrocarbons, and alcohols
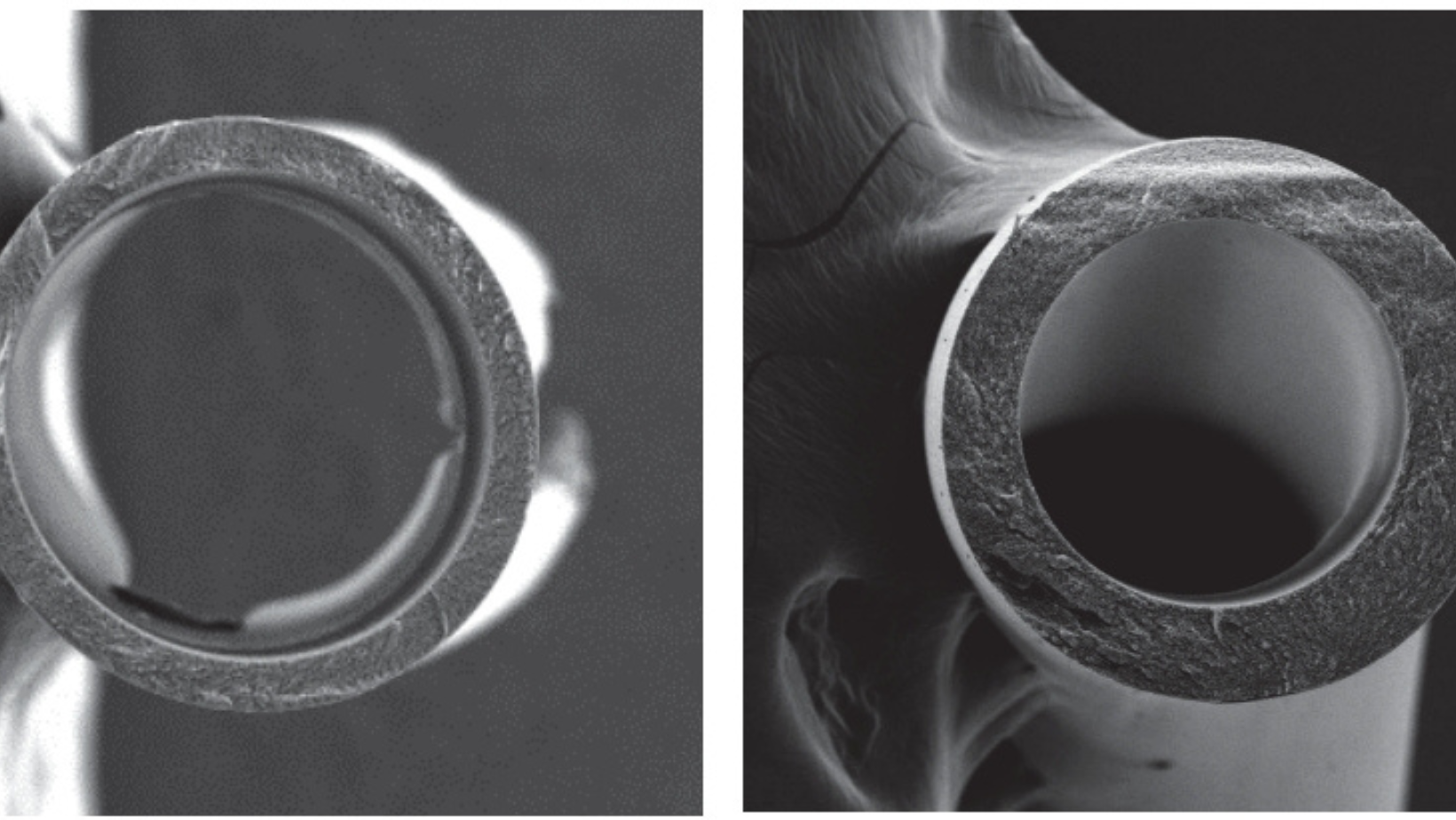
- Membrane versatility: Used in hollow fiber or composite membrane systems

From Ethanol to Ethylene Glycol
PBI membranes perform across a wide range of solvent systems.
In ethanol dehydration, PBI membranes can achieve purity levels above 99.5%, bypassing azeotropic limits.
In ethylene glycol systems, PBI maintains stability and performance where traditional membranes lose efficiency due to viscosity and temperature.
Engineering for Selectivity and Flux
Membrane performance depends on balancing flux and selectivity.
PBI hollow fiber membranes can be engineered to optimize both.
- Thickness control: Adjusts flux and rejection performance
- Thermal annealing: Improves selectivity
- Chemical crosslinking: Enhances solvent resistance and flexibility
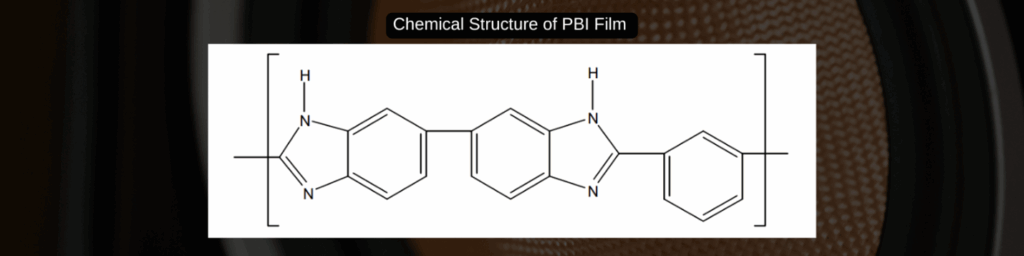
PBI vs Conventional Membrane Materials
| Property | PBI | Polyimide | P84 |
|---|---|---|---|
| Thermal Stability | Excellent | Moderate | Moderate |
| Solvent Resistance | High | Moderate | Moderate |
| Durability | High | Lower | Moderate |
Raising the Bar in Solvent Dehydration
PBI membranes enable high-performance solvent dehydration in conditions where traditional materials fail.
PBI Performance supplies polybenzimidazole (PBI) in solution form as a foundation for advanced membrane systems used in chemical separation and purification.
Contact PBI Performance Products to discuss your separation needs or request technical support.
Frequently Asked Questions
What is pervaporation?
Pervaporation is a membrane-based separation process that removes water from solvents without boiling.
Why is PBI used in solvent dehydration?
PBI offers superior thermal stability and chemical resistance compared to conventional polymers.
Can PBI handle aggressive solvents?
Yes, PBI resists ethanol, hydrocarbons, and many industrial solvents.
How does PBI improve separation performance?
It maintains membrane integrity, enabling consistent flux and selectivity.